Continuous Wet Granulation and Fluid-Bed Drying
An introduction to a revolutionary truly continuous wet granulation and drying system


By Dr. Robin Meier and Daniel Emanuele
Continuous wet granulation and drying have long been a hot topic in pharmaceutical research and development (R&D) and industry. However, there is no truly continuous fluid-bed dryer on the market that is suitable for a long process time and that meets the quality requirements described in the present article. This article outlines the experimental testing on a new R&D system (QbCon® 1) for truly continuous granulation and drying. Special attention is paid to the dryer and the resulting granules.
The results show that very narrow and reproducible residence time distributions are achieved in the dryer, which leads to uniform drying of the granules of different size classes. The process delivers consistent quality regarding moisture and particle size of the granules over several hours. When changing the conveying speed of the granules in the dryer while maintaining all other parameters, it was possible to describe the limits for a fixed process/formulation combination, which depends mainly on the depth of the fluid bed.
The possibility to follow the process in-line makes the QbCon® 1, with its innovative drying process, an ideal device for the introduction of continuous wet granulation.
The FDA Guidance on Process Analytical Technologies (PAT) and the ICH Guidelines Q8, Q9 and Q10 on pharmaceutical development, quality-related risk management and quality systems have given the concept of continuous pharmaceutical manufacturing a major boost. The pharmaceutical industry expects many advantages from the implementation of continuous manufacturing processes [1-3].
The possibility to monitor a production process more closely by means of PAT and thereby constantly produce high quality products is the key aspect of these ideas and entails many other advantages such as higher patient safety, lower costs, better process understanding and enhanced process control, to name but a few.
It is intrinsic to continuous processes that, to a certain extent, they require only longer production times instead of a scale-up, but since all developments start in the laboratory, there is a need for space-saving equipment in laboratory size, which is also suitable for the smallest throughputs.
Granulation and granule drying also play a major role in continuous manufacturing, since the reasons for granulation (improved flowability, prevention of segregation, dust reduction, etc.) are not necessarily eliminated by continuous manufacturing. However, there is currently no continuous dryer on the market that can meet the quality requirements of pharmaceutical production.
 First of all, a short residence time of the granules in the dryer with a narrow residence time distribution is essential to be able to remove defective products in a targeted manner without having to discard a large portion of the batch, which would be the case with a high degree of back-mixing. Furthermore, narrow residence time distributions simplify the traceability of material in the continuous process, making it easier and less complex to trace finished tablets back to the raw materials or to subsequently trace raw materials to the finished tablet.
First of all, a short residence time of the granules in the dryer with a narrow residence time distribution is essential to be able to remove defective products in a targeted manner without having to discard a large portion of the batch, which would be the case with a high degree of back-mixing. Furthermore, narrow residence time distributions simplify the traceability of material in the continuous process, making it easier and less complex to trace finished tablets back to the raw materials or to subsequently trace raw materials to the finished tablet.
Further important quality requirements for a dryer are even, reproducible drying of all granules – especially of different sizes – over the entire process duration, a small process chamber to keep the volume in the drying process as low as possible and, in particular, a long service life of the exhaust air filters without having to interrupt the drying process or even replace the filters.
There are several semi-continuous or quasi-continuous dryers (with several drying chambers working in parallel) for granules by different suppliers on the market, but none of them meets all the quality requirements mentioned above. In addition, semi-continuous dryers cannot deliver the promised benefit of reducing waste products that do not meet specifications to the extent of a fully continuous dryer. Due to the high volume of material that remains in different chambers of the semi-continuous drying process, it is not possible to discard specific partial quantities. On the contrary, entire partial batches must be discarded in case of doubt due to the high degree of (back-) mixing.
The present study describes and examines a novel, truly continuous dryer that is designed to combine all the above-mentioned quality requirements placed on such a system. The dryer is part of the QbCon® 1, a system for R&D, which for the first time allows the fully continuous production of granules through wet granulation and drying on a laboratory scale within a fully integrated mechanical and automated unit.
In an endurance test, the functioning of granulation and drying is investigated and the suitability for truly continuous operation is demonstrated. For this purpose, critical quality attributes of the granules are analysed over the course of the process, and the process is characterized with regard to its limits.
Materials & Methods
Materials
A powder mixture (proportions: 80/17/3) consisting of α-lactose monohydrate (Granulac 200, Meggle, Wasserburg, Germany), microcrystalline cellulose (Avicel PH 101, FMC BioPolymer, Philadelphia,PA, USA) and povidone (Kollidon 30, BASF, Ludwigshafen, Germany) was used for granulation. Purified water was used as granulation liquid.
The blue, water-soluble dye FD&C Blue No.1 (BASF) was used to assess the residence time distribution of the granules in the dryer. Manufacture of the granules / System description:
QbCon® 1
The three powders were mixed in a laboratory blender (LM 40, L. B. Bohle Maschinen + Verfahren GmbH, Ennigerloh, Germany) at a speed of 15 rpm. Granules were produced in a fully continuous process. The QbCon® 1 (Fig. 1) (L. B. Bohle) is a system with fully integrated software and unit operations consisting of different components by L. B. Bohle (liquid feeding, wet granulation, drying, automatization) and Gericke (powder feeding). Feeding of powder and liquid, wet granulation and drying are possible in one fully continuous process step without interruptions.

The entire process is controlled and monitored via a touch panel. The nominal throughput of the system is 0.5-2.5 kg/h, but can significantly deviate upwards, for example if high-density material is used, the moisture required for granulation is low or the desired residual moisture is in a higher range.
In the present tests, the powder mixture described above was filled into the gravimetric powder feeder (GZD150.12 special, Gericke, Regensdorf, Switzerland) and dosed at a rate of 1 kg/h into the first zone of the QbCon® 1 twin-screw granulator.
The granulation liquid was fed into the second zone of the granulator via a high-pressure pump using a nozzle with an internal diameter of 0.25 mm. The liquid feed rate was controlled by a Coriolis flow meter at 4 g/min resulting in a granule moisture content of 24%. In a twin-screw granulation process, powder and liquid are continuously fed onto two co-rotating screws. Both components are continuously conveyed, mixed and sheared to produce granules that leave the granulator at the end.
 For an example and overview of the granulator process zone, please see Fig. 2. The twin screw granulator of the QbCon® 1, whose screws have a diameter of 25 mm and a length of 20 x diameter, was operated at a screw speed of 50 1/min. The screw configuration consisted for the most part of double flighted conveying elements. After the liquid feeding, a kneading element consisting of five individual discs with an offset angle of 45° was used, which supports the even distribution of liquid in the granules. The kneading element was 25 mm long. Shortly before the outlet, another kneading zone with the same length and the same offset angle was inserted.
For an example and overview of the granulator process zone, please see Fig. 2. The twin screw granulator of the QbCon® 1, whose screws have a diameter of 25 mm and a length of 20 x diameter, was operated at a screw speed of 50 1/min. The screw configuration consisted for the most part of double flighted conveying elements. After the liquid feeding, a kneading element consisting of five individual discs with an offset angle of 45° was used, which supports the even distribution of liquid in the granules. The kneading element was 25 mm long. Shortly before the outlet, another kneading zone with the same length and the same offset angle was inserted.

Wet granules leave the twin-screw granulator and land on the distributor plate of the continuous dryer via a direct connection, where they are immediately subjected to hot air and conveyed in the direction of the outlet by a vibration excitation. Fluidisation of the granules through the air and vibration is extremely low, just enough for horizontal conveying. The supply air temperature and the supply air volume during the tests was 80 °C and 20 Nm3/h respectively.
The conveying of the granules and the air flow are independently adjustable, so that the drying effect of the granules can be controlled via the air flow, the supply air temperature and the conveying speed. The filter surface for the exhaust air is permanently kept free by means of a cleaning process that is based on a new principle. Thus, for the first time, a continuous granulation without interrupting the process is possible. The dry granules leave the QbCon® 1 via a pneumatically operated valve and can be further analysed and processed. A prepared mechanical interface for a NIR probe or any other similar probe also allows in-line quality determination and thus control of the process based on quality (e.g. moisture, active ingredient content or pro-duct temperature) of the granules.
Residual moisture content
The residual moisture of the granules was determined based on the loss on drying of the collected samples (approx. 3 g). For this purpose, an analytical moisture analyser (HR73 Halogen Moisture Analyzer, Mettler Toledo, Columbus, Ohio, USA) was used at a drying temperature of 105 °C. The abort criterion was a mass loss of < 1 mg within 60 seconds. The analyses were performed in triplicate for each sample.
During one test, the dry granules were divided into four size classes using test sieves of sizes 1.4 mm, 1.0 mm and 0.5 mm in order to test the uniformity of drying within the various sieve fractions.
Granule size distribution
A photo-optical analyser (CPA 2-1, Haver und Boecker, Oelde, Germany) was used to investigate the granule size distribution. A sample of dried granules collected over ten seconds was fed into the measuring zone via an automatically controlled vibrating feeder, in which a line scan camera determines the granule size and automatically transmits it to the connected software for processing. For the evaluation, the 10 %, 50 % and 90 % size quantiles of the Q3 distribution were used.
Residence time distributions
The residence time distribution of the granules in the continuous dryer was measured using ExtruVis 3 (ExtruVis, Riedstadt, Germany). The device consists of an industrial camera, a ring light for constant illumination of the room to be recorded and a special software for evaluating the data. During the process, a pulse of moist granules that had previously been dyed with a blue dye was manually fed into the inlet of the continuous dryer. The coloured granules moved together with the white granules towards the outlet.
The ExtruVis camera was placed directly in front of the outlet to record the distribution of the coloured granules within the white granules. The connected software calculates a density distribution of the residence time as a function of time (E (t)) based on the colour intensity at the outlet. A detailed description of the device can be found in Meier et al. [4]. A measuring frequency of approximately 20 images per second allowed a reliable in-line determination of the residence time distribution in the dryer. The mean residence time of the granules in the dryer is calculated by halving the area under the residence time curve.
Trial design
The main focus of this study is on the drying process of QbCon® 1. Initially, a long-term test was carried out using the process data described above, in which samples were taken at regular intervals over a period of 2.5 hours, which were examined under various quality aspects.
Thereafter, the drying conditions were varied by changing the conveying speed of the granules in the dryer while maintaining all other process and formulation parameters. The purpose was to investigate the process limits of the dryer for the underlying conditions depending on the residence time.
Results and discussion
It was generally observed that the continuous granulation and drying tests could be carried out without interrupting or disturbing the process. The critical process parameters of feeding, granulation and drying were consistent over the entire test period. Particularly noticeable was that the exhaust filters were covered with only a small portion of the fines content of the granules. This low coverage can be explained by the extremely low fluidisation of the granules in the dryer. Therefore, the cleaning interval of the product filters has been reduced to 20 minutes. The filters could be cleaned completely without any product residues during cleaning.
Residual moisture is a critical quality attribute for dried granules. Highly fluctuating values and too high residual moisture contents can lead to irregularities in downstream processes, e.g. smearing of sieve surfaces, variations in tablet content or difficulties with the continuous feeding of granules. The residual moisture of the produced granules after continuous drying over a test period of 2.5 hours is shown in Fig. 3. The raw material had an initial moisture content of 1.28 %. The residual moisture of the granules fluctuated between 1.02 % and 1.38 % during the test and averaged 1.21 ± 0.11 %. This scattering around the mean value is acceptable for residual moisture measurement by means of LOD and can be regarded as very low.
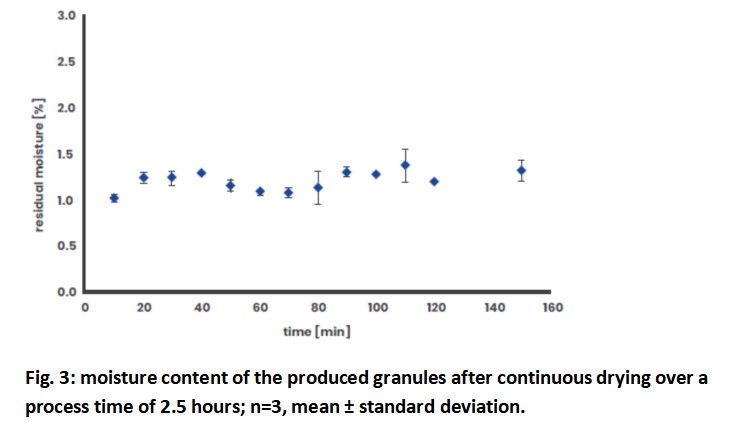
With regard to the moisture content of the dried granules, a consistent and reproducible result could be obtained, which is of utmost importance for a continuous process. The drying was so effective and efficient that some of the granules showed a moisture content which was below that of the raw material. This can be of great importance for moisture-sensitive products. If the residual moisture is considered too low, it is possible to adapt the process to shorter drying times (= residence times), lower temperatures and air flow rates in order to achieve higher residual moisture contents.
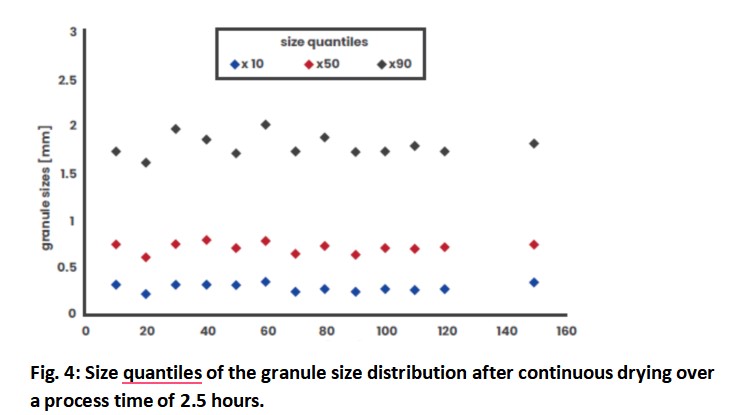
In addition to residual moisture, a consistent granule size distribution is important for further processing of the granules. The 10, 50 and 90 % quantiles of the granule size distributions are shown in Fig. 4. Again, the continuous granulation and drying process leads to consistent granule sizes, which is mainly due to the low fluctuations in the 10 % quantile (mean value: 298 μm) and in the median granule size (mean value: 726 μm). Fluctuations in the 90 % quantile are typical for twin-screw granulation and are the highest in absolute terms within these tests, but really the smallest with a relative standard deviation of 6 %. Thus, the fluctuations in the 90 % quantile (mean value: 1805 μm) are acceptable. In addition, the oversized granules are usually classified after the drying step in order to obtain a narrower size distribution.
 The residence time of the granules in the dryer defines the time in which the moist granules are exposed to the drying air flow. A constant residence time distribution is therefore of utmost importance for consistent drying results. Even though residual moisture and granule sizes indicate a constant residence time, a detailed investigation of the residence time behaviour in the continuous dryer is of great interest, both for upscaling to larger systems and for discharge concepts for faulty material in fully continuous processes.
The residence time of the granules in the dryer defines the time in which the moist granules are exposed to the drying air flow. A constant residence time distribution is therefore of utmost importance for consistent drying results. Even though residual moisture and granule sizes indicate a constant residence time, a detailed investigation of the residence time behaviour in the continuous dryer is of great interest, both for upscaling to larger systems and for discharge concepts for faulty material in fully continuous processes.
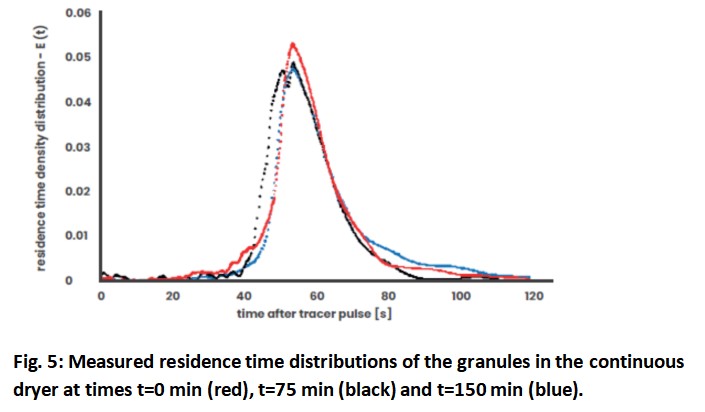
Therefore, the distribution of the residence time of the granules in the dryer at the beginning, middle and end of the endurance test is shown in Fig. 5. It is not surprising that the curves are almost congruent, as the results discussed above have already indicated this. The mean residence time for the three tests was 57, 61 and 63 seconds, which is very short for continuous drying. The fact that such low residual moisture is still maintained, demonstrates the effective flow of the drying air through the granules. Not only the mean residence time, but also the width of the residence time distribution is an important parameter that characterizes the conveying and blending behaviour of a system. The small width of the measured distributions is ideal for a continuous dryer. The aim must be to move all granules through the dryer at the same speed in order to achieve a similar drying performance. Wide residence time distributions can lead to different moisture content within a sample. The curves are almost axis-symmetrical, which indicates very low back-mixing in the dryer.
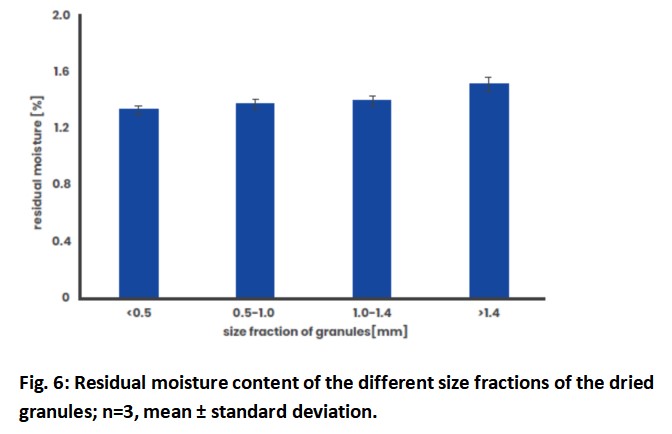
Due to these considerations, the uniformity of the drying process within a sample must be tested. Optically, the observations did not reveal any differences between the conveying speed of coarse and fine particles. Fig. 6 shows the residual moisture of granules of different sizes from the same sample, which show only very small differences. Only the granules of the largest fraction have a slightly higher moisture content, which is common with fluid-bed drying and is of no greater significance within the scope shown here.
In order to further characterize the QbCon® 1 and to test the limits for the present formulation and throughput, the conveying speed of the granules was increased or decreased by adjusting the vibration feeder. This results in different granule bed heights in the dryer and residence time distributions varying in length and width, which are shown in Fig. 7. The mean residence times vary between 49 and 229 seconds and the curves show a high degree of symmetry. The residence time distribution with the longest mean residence time is particularly striking. The long-time interval between the first and last occurrence of coloured granules in the residence time experiment suggests that the granules of a sample are unlikely to become uniformly dry.
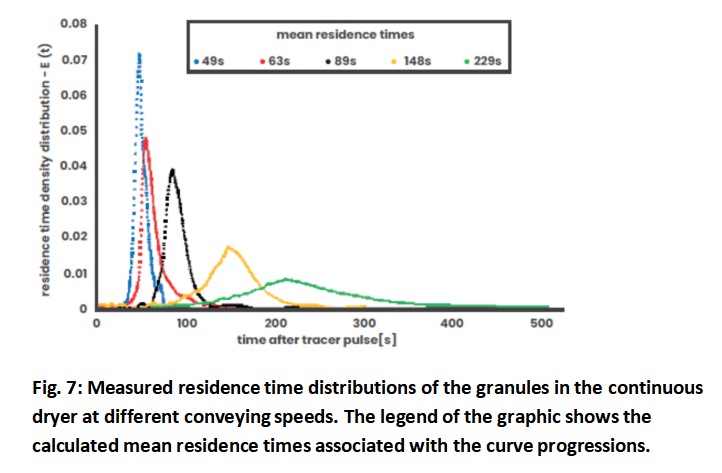
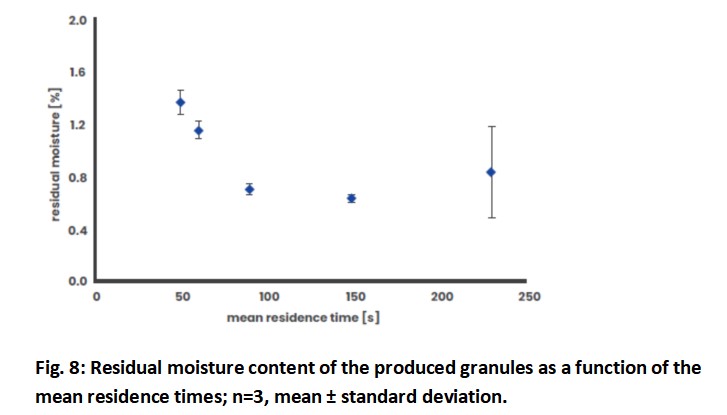
 This is illustrated by Fig. 8, where the residual moisture of the granules is displayed as a function of the mean residence time. With the mean residence time increasing from 49 to 148 seconds, the residual moisture of the granules drops from 1.37 to 0.65%. However, if the mean residence time is increased to 229 seconds and the residence time curve is widened at the same time, the residual moisture will increase again. In addition, the values scatter very widely with a relative standard deviation of > 41 %. If the residence time is too long, the bed thickness of the granules in the dryer increases, resulting in a poorer and less even flow through the granules. These two influencing parameters are of utmost importance when designing the desired process.
This is illustrated by Fig. 8, where the residual moisture of the granules is displayed as a function of the mean residence time. With the mean residence time increasing from 49 to 148 seconds, the residual moisture of the granules drops from 1.37 to 0.65%. However, if the mean residence time is increased to 229 seconds and the residence time curve is widened at the same time, the residual moisture will increase again. In addition, the values scatter very widely with a relative standard deviation of > 41 %. If the residence time is too long, the bed thickness of the granules in the dryer increases, resulting in a poorer and less even flow through the granules. These two influencing parameters are of utmost importance when designing the desired process.

Conclusion
A new system for the continuous production and drying of granules on a laboratory scale was experimentally investigated. The present tests have shown the great potential of this novel drying process which uses a fluid bed. Thanks to a very short residence time and a narrow residence time distribution, constant, continuous drying of the granules was achieved even among the different size classes, and the particle size distributions were also constant over the process time. Such results are so far unique in the field of continuous wet granulation and demonstrate the superiority of the present system over known processes.
On the basis of different conveying speeds of the granules in the dryer, it was possible to determine the process limits for a given formulation and a given throughput – simply based on the residence times and residual moisture content. The height of the granule bed proved to be a critical parameter to be controlled. The possibility to follow the process in-line makes the QbCon® 1, with its innovative drying process, an ideal device for the introduction of continuous wet granulation within companies.
The possibility to follow the process in-line makes the QbCon® 1, with its innovative drying process, an ideal device for the introduction of continuous wet granulation within companies.
Literature
[1] H. Leuenberger, New trends in the production of pharmaceutical granules: Batch versus continuous processing, Eur. J. Pharm. Biopharm., 52 (2001) 289-296.
[2] C. Vervaet, J.P. Remon, Continuous granulation in the pharmaceutical industry, Chem.Eng. Sci., 60 (2005) 3949-3957.
[3] J. Rantanen, J. Khinast, The future of pharmaceutical manufacturing sciences, J.Pharm. Sci., 104 (2015) 3612-3638.
[4] R. Meier, M. Thommes, N. Rasenack, K. P. Moll, M. Krumme, P. Kleinebudde, Granule size distributions after twin-screw granulation – do not forget the feeding systems, Eur. Journ.Pharm. Biopharm., 106 (2016), 59-69.
ABOUT THE AUTHORS
Dr. Robin Meier is Manager Scientific Operations at L.B. Bohle Maschinen und Verfahren GmbH and in his function responsible for the Technology Center, the QbCon®-line at L.B. Bohle and all scientific collaborations with universities and within scientific consortia. Robin is a pharmacist by education and received his doctorate in pharmaceutical technology and biopharmacy with his work on the continuous production of tablets based on twin-screw granulation. He is leading an experienced team of process- and automation engineers, who are focusing on the continuous improvement and development of the existing batch machines and the continuous manufacturing equipment.
Daniel Emanuele studied Bioengineering with a focus on Process Engineering at the Technical University of Dortmund. For his experimental thesis, he investigated the influence of powder transfer on the production of moist granules within the QbCon 2.5 continuous production plant from L.B. Bohle. Since 2018, he has played a key role in commissioning and supporting the QbCon 1.

