Automation and Robotics in Pharma Packaging
Towards Zero Human Error

Pharmaceutical packaging is more than a protective shell; it is a promise of safety, efficacy, and integrity. As global health demands rise and regulatory scrutiny intensifies, packaging operations have emerged as a strategic focus area for the pharmaceutical industry. The challenge is no longer limited to sealing tablets or labelling vials; it is about achieving consistency, compliance, and efficiency – all while minimizing the possibility of human error.
Over the last decade, automation and robotics have redefined how packaging floors operate. What was once a labour-intensive process has evolved into a highly orchestrated, data-driven system that combines precision engineering, artificial intelligence, and real-time monitoring. This transformation has not only increased output but has also brought the industry closer to its ultimate goal – zero human error.
The Evolution of Pharma Packaging Automation
The Manual Era
In the early years, pharmaceutical packaging was largely manual. Operators filled bottles, inserted leaflets, and sealed cartons by hand. Inspection relied heavily on human eyesight. Errors, though unintended, were frequent – wrong labelling, missing leaflets, or incorrect batch printing. Productivity was limited by human fatigue and attention span.
Semi-Automation and the Age of Conveyors
By the late 1990s and early 2000s, semi-automatic machines and conveyors began transforming the production floor. These machines reduced repetitive tasks and improved throughput, but still required constant human intervention for setup, material loading, and inspection.
The Era of Full Automation
The 2010s marked the beginning of fully automated packaging lines. Programmable Logic Controllers (PLCs), servo-driven systems, and sensors replaced manual judgment with precise, repeatable movements. The focus shifted from “doing the work” to “monitoring the system.”
The Rise of Robotics and Smart Manufacturing
Today, the integration of collaborative robots (cobots), vision systems, digital twins, and AI-based inspection has redefined packaging excellence. The modern packaging floor resembles a digital ecosystem – responsive, self-diagnosing, and interconnected.
Key Drivers behind Automation
Regulatory Compliance
Strict global regulations (GMP, FDA, EU Annex 11, 21 CFR Part 11) demand traceability and data integrity. Automation ensures every operation is logged, validated, and reproducible.
Demand for Speed and Flexibility
The rise of personalized medicine and smaller batch sizes requires flexible systems that can handle multiple SKUs, packaging formats, and serialization codes without prolonged changeovers.
Cost and Labour Efficiency
Skilled labour shortages and rising costs are driving companies to automate repetitive and high-risk operations. Automation not only reduces dependency but also ensures consistency across shifts and locations.
Safety and Ergonomics
Replacing manual tasks with robotics reduces operator fatigue, musculoskeletal stress, and exposure to dust or chemicals, promoting a safer working environment.
Components of a Modern Automated Packaging Line
A fully automated packaging line integrates several subsystems, each communicating through a central control architecture.
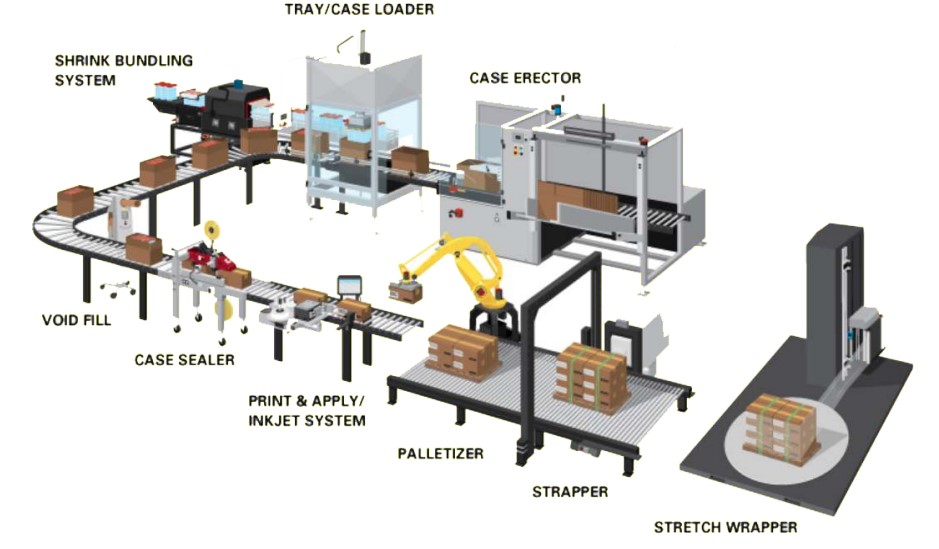
Feeding Systems
Automated feeders ensure the right count and orientation of products entering the line. Vibratory bowl feeders, pick-and-place robots, and intelligent conveyors align and deliver tablets or vials precisely.
Blister and Bottle Packaging
Advanced blister machines employ servo motors for accurate forming, sealing, and punching. In bottle packaging, automatic counting, desiccant insertion, and capping ensure uniformity at high speeds.
Cartoning and Case Packing
High-speed cartoners handle leaflets and cartons with robotic precision. Case packers assemble multiple cartons into secondary packages seamlessly, often integrating serialization and aggregation systems.
Vision Inspection Systems
Machine vision systems use high-resolution cameras and AI algorithms to detect defects, misprints, or missing components far more reliably than the human eye.
Labelling and Serialization
Automatic labelling machines integrate with global track-and-trace databases, applying unique serial numbers and verifying them in real time to combat counterfeiting.
Palletizing and Dispatch
End-of-line palletizing robots stack boxes based on predefined patterns, reducing manual handling and improving warehouse safety.
Robotics in Packaging: From Assistance to Autonomy
Robots have evolved from fixed pick-and-place arms to intelligent, self-learning assistants capable of complex decisions.
Collaborative Robots (Cobots)
Cobots work safely alongside humans without cages. They handle repetitive tasks like carton loading or labelling while operators focus on quality monitoring and decision-making.
AGVs and AMRs
Automated Guided Vehicles (AGVs) and Autonomous Mobile Robots (AMRs) transport materials between production and packaging zones, ensuring uninterrupted flow and minimizing human movement.
Robotic Inspection
Equipped with 3D vision and AI, robots can perform inline quality checks –inspecting fill levels, verifying caps, or reading barcodes – faster and with greater accuracy than manual inspectors.
Self-Learning Systems
Machine learning enables robots to adapt to variations in packaging components. For instance, if a foil slightly changes in texture or reflectivity, AI algorithms adjust inspection thresholds automatically.

Human Error and the Pursuit of Zero Defects
Human errors in packaging can have serious implications – from regulatory recalls to patient harm. Automation minimizes these risks by eliminating subjectivity.
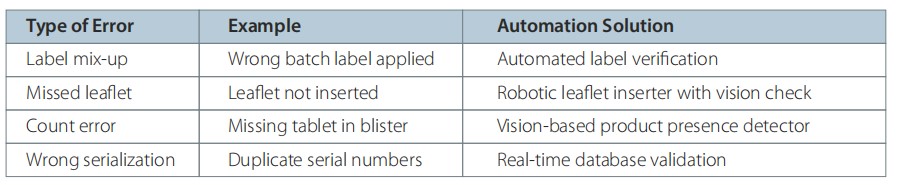
Automation ensures “error-proofing” (Poka-Yoke) through interlocks, barcode scanners, and feedback loops that stop the process immediately when deviations occur.

Data, Digitalization, and Smart Analytics
Modern packaging systems are no longer mechanical islands. They generate, analyze, and transmit data continuously.
SCADA and MES Integration
Supervisory Control and Data Acquisition (SCADA) and Manufacturing Execution Systems (MES) connect machines with enterprise-level data platforms, providing real-time visibility of every batch.
Predictive Maintenance
Sensors monitor vibration, temperature, and torque, predicting machine wear before a breakdown occurs – minimizing downtime.
Digital Twin Technology
A digital twin is a virtual replica of the packaging line. Engineers simulate setups, run diagnostics, and optimize performance without stopping the physical line.
AI-Driven Process Optimization
Artificial intelligence identifies bottlenecks and automatically suggests speed or parameter adjustments, making the system self-optimizing.
Benefits of Automation and Robotics
Consistency and Accuracy – Each operation follows a predefined algorithm, reducing variability.
Higher Throughput – Multi-head robotics achieve production speeds unimaginable for manual setups.
Regulatory Readiness – Data integrity and electronic records simplify audits and validation.
Operational Safety – Automation isolates humans from high-speed or repetitive zones.
Energy and Material Efficiency – Precise dosing and reduced wastage lower carbon footprint.
Challenges and Considerations
High Initial Cost
Investment in robotics, sensors, and data infrastructure can be significant, but the long-term savings in labour, rejects, and downtime justify it.
Skill Gap
Operators must be retrained in mechatronics, PLC programming, and system analytics – a paradigm shift from manual supervision.
Integration Complexity
Combining machines from multiple vendors requires robust communication protocols (e.g., OPC-UA, MQTT) to ensure seamless data exchange.
Cybersecurity
With connectivity comes vulnerability. Packaging lines need firewalls, user authentication, and encrypted communication to prevent data breaches.
The Future: Towards Lights-Out Packaging
The concept of a “lights-out” packaging line – operating autonomously 24/7 with minimal human presence – is no longer science fiction. With advancements in robotics, AI, and IoT, pharma packaging is moving towards self-healing systems that can detect, analyze, and correct faults on their own.
Integration with AI and IoT
IoT-enabled sensors feed data to centralized dashboards. AI algorithms interpret this data to make micro-decisions – adjusting seal temperature, conveyor speed, or torque dynamically.
Sustainability and Green Packaging
Automation supports sustainability through precision – reducing film waste, optimizing carton sizes, and monitoring energy consumption in real time.
Personalized Packaging
Future systems will adapt packaging configurations automatically based on product profiles or patient-specific regimens, aligning with the era of personalized medicine.

The Human Role in an Automated World
Even in a highly automated environment, humans remain central – as designers, analyzers, and decision-makers. Automation reduces drudgery, allowing professionals to focus on innovation and problem-solving.
Training programs must evolve to prepare operators for the new digital workplace – focusing on data interpretation, maintenance diagnostics, and cross-disciplinary understanding of mechanical and IT systems.
Conclusion
Automation and robotics have not merely improved pharma packaging – they have transformed it. The journey from manual handling to self-learning systems mirrors the industry’s broader evolution toward precision, safety, and intelligence.
As the pharmaceutical world advances toward Industry 5.0, the collaboration between human intuition and machine precision will define packaging excellence. The goal is not to replace people but to empower them – enabling smarter, faster, and safer operations that deliver medicines with absolute confidence and zero human error.

ABOUT THE AUTHOR
Dr. Pritam Rajkumar Siraskar (M. Pharm., Ph.D.) is an accomplished pharmaceutical professional, academic mentor, and industry leader with more than 15 years of extensive experience across pharmaceutical manufacturing, quality management, and research.
He is currently serving as Senior Manager and Head of Production at Glenmark Pharmaceuticals, Indore, India. Dr. Siraskar holds a Master of Pharmacy degree from RGPV, Bhopal, and a Ph.D. in Pharmaceutical Sciences. His professional expertise spans oral solid dosage forms (OSD), liquids, semisolids, dry syrups, and suppositories, along with technology transfer, research and development, and the successful execution of greenfield and brownfield pharmaceutical projects.
Dr. Siraskar has authored multiple research publications and 4 books and several book chapters in reputed national and international journals and academic volumes. He is actively engaged in academic mentorship and is currently guiding six Ph.D. research scholars in the field of pharmaceutical sciences.
A Certified Six Sigma Black Belt and Green Belt, Dr. Siraskar also holds PMP and PMI certifications, reflecting his strong foundation in process excellence and project management. Throughout his career, he has collaborated with several leading pharmaceutical organizations, including Ranbaxy, Intas, Mylan, Wockhardt, Hetero, Cipla, and Glenmark Pharmaceuticals.
Deeply committed to lifelong learning and professional development, Dr. Siraskar has successfully completed over 1,000 professional certifications across technical, regulatory, quality, and managerial domains. His work continues to focus on strengthening industry–academia collaboration, promoting research quality, and mentoring the next generation of pharmaceutical professionals.

