Fungi in Cleanrooms : Types, Origins, and Decontamination Strategies
By Tim Sandle

Cleanrooms and controlled environments are at risk from fungal spores, should they enter the facility (1). Fungi can present health hazards to operators and pose potential contamination risks to medicinal products. The presence of fungi in the processing areas of pharmaceutical and healthcare products manufacturing facilities is normally regarded as an indication of a control breakdown, such as one relating to air handling or with the transfer of materials (or people) into the manufacturing area. The risk exists because most fungi produce spores, for reproduction or for survival, and such spores are relatively easy to disperse in the form of bioaerosols. These biologically dynamic particles can cover relatively wide distances and, unlike bacterial spores, fungal spores are found in relatively high numbers. The risk of spore dispersal can be enhanced based on airflow dynamics, the ambient temperature and humidity, plus water availability.
This article considers the types of fungi that might be recovered from cleanrooms and looks at the mechanisms by which they might enter a facility. The article concludes with some considerations for fungal remediation.
Types of fungi recovered from cleanrooms
Fungi are structurally complex organisms, showing a variety of reproductive structures associated with sexual and asexual processes, in addition to vegetative, nonreproductive elements and hyphal structures. Their differentiation into genera, species, and varieties is made, in a large part, on a morphological basis — especially morphology of the reproductive structures.
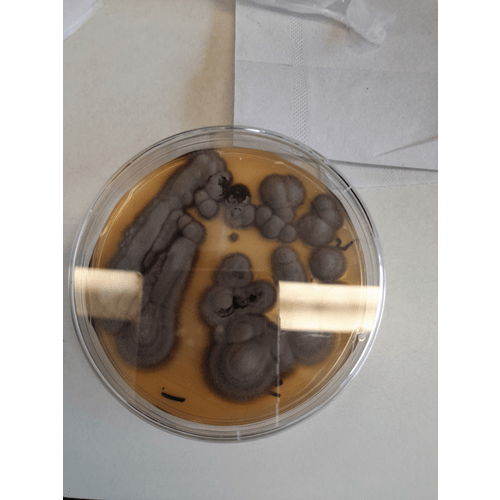
There is a huge diversity of fungi on the planet. Mycologists have identified about 100,000 to 120,000 species of fungi, yet this 100,000 or so is only a fraction of the 1.5 million species of fungus probably present on earth.New culture-independent approaches to determine microbial diversity, such as next-generation sequencing methods, are greatly broadening our view of fungal diversity (2). The number of different fungal species associated with the as-built environment is somewhere between 200 and 250, with the majority requiring aerobic conditions for growth (3).
While there are geographical variations in terms of what might be isolated (4), the species of Aspergillus, Cladosporium, Penicillium, Alternaria, Curvularia and Fusarium are the most predominant fungal isolates from pharmaceutical processing environments (based on a review undertaken by this author) (5). These fungi present a hazard and a challenge to contamination control practices. This is not least because spores of fungi can be found in dustparticles in the atmosphere or on floors, worksurfaces or equipment.
Fungi come from many sources in the cleanroom, including door kick plates, bags, incubators, boxes, markers, intervention equipment, cartwheel, ceiling tiles, poorly maintained flooring, vibrations from construction, and in some cases, high-pressure impingement application devices for applying cleaning solutions.
How fungi enter cleanrooms
Fungi come from many sources in the cleanroom, including door kick plates, bags, incubators, boxes, markers, intervention equipment, cartwheel, ceiling tiles, poorly maintained flooring, vibrations from construction, and in some cases, high-pressure impingement application devices for applying cleaning solutions. Deficient room pressurization, vibrations from construction, poor engineering designs related to sprinkler systems and light fixtures have also been the cause of fungal outbreaks in cleanrooms. Variations with the environment, such as increases in ambient temperatures, together with items brought into cleanrooms, are also contributors.
Overall, there are probably two main types of breaches: the ingress of unfiltered air (where atmospheric air poses the greatest risk), such as from faulty Heating, Ventilation and Air Conditioning (HVAC systems); and with the ingress of water, such as a leak from a pipe (6). Other factors include transfer via personnel, where gowning can present a particular concern (the toe area has the highest population of fungi on the human body and the greatest varieties of fungi are found on the heel of the foot) (7). These points of origin are revisited in the section below about prevention of fungal contamination.

Fungal spores and dispersal
Fungal spores are the reproductive mechanism for fungi. Fungal spores are either unicellular or multicellular, developing into a number of different phases through the complex life cycles of the fungi.
For example, with filamentous fungi the process of growth is through the production of hyphae (tubular branches), which expand as a mass (called the mycelium) in multiple directions. The production of aerial hyphae is an important mechanism for spore dispersal (8). In the cleanroom environment, the physical determinants in relation to each Heating, Ventilation and Air Conditioning (HVAC) unit will introduce air vector variabilities.
In terms of the way spores may be distributed within the cleanroom environment, with either sexual or asexual reproduction, the spores that disperse from the parent organism will do so either by either air currents or hitching a ride on a moving person or object. As well as dispersal mechanisms of the fungus itself, spores can move by water droplets. In terms of settling, sedimentation at terminal velocity under gravity is an important factor. Where spores are kept in suspension and removed from the controlled environment, the risks are lower; where spores settle the risk of germination exists, depending on those confluence of the necessary factors designed to promote germination (as discussed in this chapter). With the sedimentation of fungal spores, different types of spores do not equally remain in air. The deposition of a given spore is influenced by their terminal velocity and aerodynamic characteristics. In general, large conidia and spores will settle faster than smaller ones.
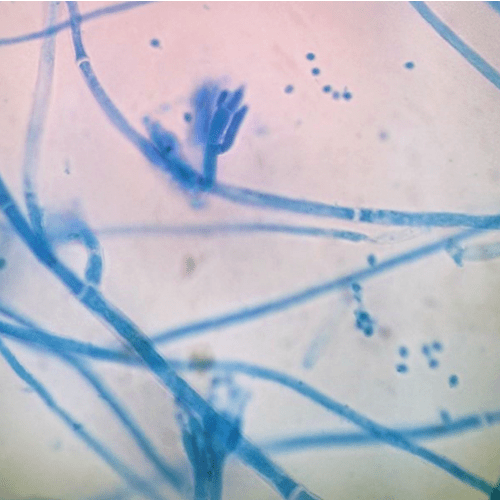
The objective of most fungal spores is reproduction. Fungi are often classified according to their spore-producing structures, for example, spores produced by an ascus are characteristic of ascomycetes. Different factors will influence the germination process, especially temperature which has a marked effect on the spherical growth rate of spores and their eventual size. The temperature will need to be optimal for the fungal species. A second important factor is with humidity. Germination is also mediated by surface topography, hydrophobicity, and physical and chemical signals (9). If conditions are not favorable, such as a low relative humidity, the spore will remain in dormancy (10).
As well as reproduction, some fungi produce spores to survive unfavorable environmental conditions (such as hot and dry environmental conditions, lack of nutrients, alterations to osmolarity and hostile pH levels and chemicals), these are termed chlamydospores (a chlamydospore is the thick-walled large resting spore of several kinds of fungi.
Why fungi survive in cleanrooms
Upon entry of viable fungal propagules from outdoors, the fungus most probably becomes attached to a susceptible substrate (11). If fungi enter the cleanroom, the prevailing conditions of temperature, water availability and the state of the fabric are key determinants of whether the fungi will survive and colonize. If colonization occurs, it tends to follow the pattern: vegetative growth will commence if the appropriate environmental conditions exist. These conditions include sufficient free water and a susceptible substrate that can provide the necessary nutrients for fungal growth. If the conditions are suitable, the fungus will release extracellular fungal enzymes into the immediate environment, from which nutrients are absorbed and for which the presence of water is important (12).
Overall, there are probably two main types of breaches: the ingress of unfiltered air (where atmospheric air poses the greatest risk), such as from faulty Heating, Ventilation and Air Conditioning (HVAC systems); and with the ingress of water, such as a leak from a pipe. Other factors include transfer via personnel, where gowning can present a particular concern (the toe area has the highest population of fungi on the human body and the greatest varieties of fungi are found on the heel of the foot).
The indoor accumulation of water is the primary predisposing factor for indoor fungal colonization (and the subsequent biodeterioration of building materials). This is why fungi can regularly be recovered from wet-areas and wash-bays (and consequence the controls leading into and out of these areas are critical). Water is the solvent that needed by all fungi in order to grow (while some fungi can survive with very low levels of water activity (xerophilic organisms), the typical lower level for most species is an aw of 0.80 – examples include species of Aspergillus and Penicillium, which can grow slightly below 0.80; and species of Altemaria and Cladosporium which prefer the range 0.8 to 0.90 aw) (13). Water activity (aw) is different to water availability and it refers to “free” water available for microbial growth.
The objective of most fungal spores is reproduction. Fungi are often classified according to their spore-producing structures, for example, spores produced by an ascus are characteristic of ascomycetes. Different factors will influence the germination process, especially temperature which has a marked effect on the spherical growth rate of spores and their eventual size.
Preventative measures
There are several preventative measures to reduce the likelihood of fungi being present in cleanrooms. These are (14, 15):
- Control the delivery of items into the cleanrooms, such as ensuring items are dry and multi-wrapped (and that a layer is removed prior to entering the cleanroom, together with a disinfection activity). Ensure that cardboard never enters the cleanroom.
- Control of air, ensuring that the room HEPA filters are functioning as designed. One common source of fungi is the interior surface of air ducts.
- Ensure good airflow within the cleanroom. Many cleanrooms are designed as turbulent flow areas, they can move fungal spores over a wide area. The success or failure of the cleanroom design, including the ability to keep particles suspended and the number of air exchanges (determining how quickly air is removed from the cleanroom) can influence whether fungal spores settle onto new surfaces.
- An important determinant is with the materials used for cleanroom construction; while modern cleanrooms are constructed of materials that are not intended to encourage fungal growth, this is not always the case with aging facilities).
- Maintaining areas in a dry state is important. As dry states prevent fungal growth (although fungi will often survive they are less likely to reproduce).
- Minimize dust levels. Dust presents an interesting substrate, and it plays a role in spore dispersal dust can sometimes contain the nutrients derived from dead microbes, providing a nutrient source for the organisms.
- Fungi can be carried in by personnel, therefore effective gowning must be practiced.
- Use effective disinfectants. The variations between different species of fungi can be shown through assessments of the Minimum Inhibitory Concentration (MIC) of different biocides.The disinfectant used must have proven properties against a range of fungi. Often different contact times are required to eliminate spores (fungal and bacterial) than are used for standard disinfection cycles.To kill spores – both bacterial and fungal – a sporicide needs to be used. As well as killing spores, a sporicide is expected to also kill all vegetative cells. Examples of suitable disinfectants are ones that is chlorine based, or peracetic acid based, or hydrogen peroxide, or a blend of peracetic acid and hydrogen peroxide).
Assessing the presence of fungi in pharmaceutical cleanrooms requires an effective environmental monitoring regime that is capable of detecting fungi consistently. The frequency of monitoring should be sufficient as to allow trending to take place. In terms of detection, thought needs to be given to the culture medium (either a general agar designed to recover bacteria and fungi, or a specific mycological medium) and the incubation conditions (16). Giving thought to this is important since there is an association between the failure to detect fungi in the environment and the contamination of healthcare products (17).
Conclusion
Fungi pose a risk to pharmaceutical cleanrooms, given their ability to survive for long periods of time and their rapid growth once conditions are optimal (especially given the right levels of water activity). In addition, fungi can be easily spread in the air, where the different parts of the fungus can serve as reproductive propagules: conidia (asexual), spores (asexual or sexual), and hyphal fragments. Each of these can establish growth at a new nutrient source. In addition, spores often have the ability to remain dormant for substantial time periods. Due to the risks presented by fungi, this means that assessing fungal populations in cleanrooms becomes an important part of the environmental monitoring program using both air and surface methods. Once fungi have been detected in high numbers, steps need to be taken to eliminate the population. This generally relates to eliminating water sources; repairing damage; and effectively disinfecting the area.
This article has considered some of the contamination control and fungal spore risks within controlled environments. Understanding these risk factors will help to construct a contamination control strategy.

Tim Sandle is the author of the book Digital Transformation and Regulatory Considerations for Biopharmaceutical and Healthcare Manufacturers, Volume 1: Digital Technologies for Automation and Process Improvement, available via the PDA Bookstore: https://www.pda.org/bookstore/product-detail/5897-digital-transformation-volume-1
References
- Sandle, T. (2014) Fungal contamination of pharmaceutical products: the growing menace, European Pharmaceutical Review, 19 (1): 68-71
- Kousser, C., Alam, F. and Hall, R. (2019) Fungal interactions in health and disease, Microbiology Today, 46 (2): 95-97
- Flannigan B, Samson RA, Miller JD. (2002) Microorganisms in Home and Indoor Work Environments: Diversity, Health Impacts, Investigation and Control. London: Taylor and Francis
- Taylor JW, Turner E, Townsend JP, Dettman JR, Jacobson D. (2006) Eukaryotic microbes, species recognition and the geographic limits of species: examples from the kingdom Fungi. Philos Trans R Soc Lond B Biol Sci. 361:1947–1963
- Sandle, T. (2011): A Review of Cleanroom Microflora: Types, Trends, and Patterns, PDA Journal of Pharmaceutical Science and Technology, 65 (4): 392-403
- Sandle, T. (2018) Investigating and Addressing Fungal Contamination in Pharmaceutical Cleanrooms, Journal of GxP Compliance, 22 (1): 1-10
- Sandle, T. (2015) “From Head to Toe: Mapping Fungi across Human Skin”. EC Microbiology1: 211-213
- Wosten, H.A., Schuren, F.H., Wessels, J.G. (1994) Interfacial self-assembly of a hydrophobin into an amphipathic protein membrane mediates fungal attachment to hydrophobic surfaces, EMBO J, 13: 5848-5854
- Osherov, N. and May, G.S. (2001) The molecular mechanisms of conidial germination, FEMS Microbiol Lett, 199: 153-160
- El-Enshasy, H. A. (2007) Filamentous Fungal Cultures – Process Characteristics, Products, and Applications. In Yang, S. A. (Ed.) Bioprocessing for Value-Added Products from Renewable Resources: New Technologies and Applications, Elsevier, New York, pp225-261
- Money, N. P. (2016) The Fungi, 3rd edition, Chapter 3 – Spore Production, Discharge, and Dispersal, Academic Press, New York, pp67-97
- Cundell T (2014) Microbiology: mould contamination in pharmaceutical drug products and medical devices. Eur Pharm Rev18(3):67–75
- Cundell, A. M. (2009) Chapter 9: Effects of Water Activity on Microorganisms In Cundell, A. M and A Fontana (Editors) Water Activity Application in the Pharmaceutical IndustryDavis Horwood/PDA
- Sandle, T. (2013). ‘Dimorph and Filamentous Fungi’. In Mascellino, M. T. (Ed.) Bacterial and Mycotic Infections in Immunocompromised Hosts: Clinical and Microbiological Aspects, OMICS Group Inc.: Henderson, NV, USA E-book at: http://www.esciencecentral.org/ebooks/bacterial-mycotic-infections/pdf/bacterialmycotic-infections.pdf
- Vijayakumar, R., Kannan, V.V., Sandle, T. and Manoharan, C. (2012). In vitro Antifungal Efficacy of Biguanides and Quaternary Ammonium Compounds against Cleanroom Fungal Isolates. PDA Journal of Pharmaceutical Science & Technology, 66 (3): 236 – 242
- Sandle, T. (2014) Examination of the order of incubation for the recovery of bacteria and fungi from pharmaceutical cleanrooms. Int J Pharm Compd18, 242–247
- Ahearn D.G. and Stulting, R.D. (2014) Fungi associated with drug recalls and rare disease outbreaks, J Ind MicrobiolBiotechnol41: 1591 – 1597












