
By: Prabir K Das
The Product-Packaging-Labelling relationship is analogous to Automation-Digitalization-Communication relationship, practically inseparable from each other.
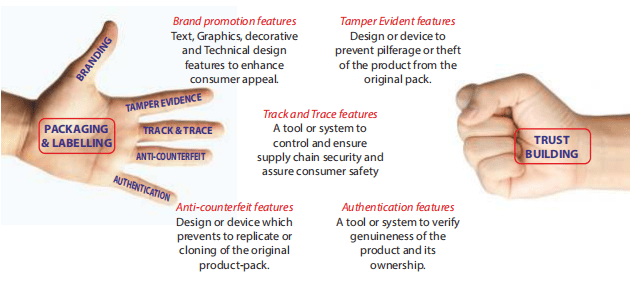
The product-people connectivity is very important in trust building process and success of a brand is largely dependent on this trust building. The packaging-labelling effectiveness is largely responsible to establish this connectivity with its communicative features. While some of the features on packaging and labelling are visible and readable, there are features which are invisible and require different tools to make it visible or readable to know the desired information.
The purpose of including these features in packaging-labelling is either to promote the brand or to protect the brand or both. The basic packaging and its in-built design prevent product spillage, leakage, breakage and spoilage. Adverse external environmental conditions and external threats like theft, diversion, and cloning are very common. Apart from internal operational controls, there are many external logistics controls too, to protect the product from these threats and challenges.
Similarly, labelling helps to comply various regulatory guidelines through instructions for identification, storage, handling, dispensing and disposal. There are elements for branding too through text, color and graphics. A combination of packaging-labelling design and features take care of all these requirements to support brand promotion and brand protection.
Over the years industry has experienced difficult situations and responded with stronger designs and features to sustain and survive in the competition. Now, in this fenceless global business, when multiple technologies are available for easing the process of globalization, industry has responded well to compete in the global market. Quantity, quality and strict regulatory complianceare the need of the hour. Evolution in industrial automation and digitalization are strongly supported with revolution in communication systems. Most of the advanced features in product-packaging-labelling became feasible because of this automation-digitalization-communication system. This is how they are analogous to each other and also inseparable from each other.
These advanced value-added features are primarily two types:
Static / standalone / off-line category – Pilfer-proof / tamper-evident feature, covert / overt anticounterfeit feature, brand promotion feature, animated and digital graphic design features, etc.
Dynamic / connecting / on-line category – Serialization / aggregation (track & trace) feature, SMS / Web based verification feature, User interactive feature, etc. Most of these features are adopted through advanced technologies – automation and digitization, wireless communication and hybrid technologies.
Brand promotion features
Text, Graphics, Decorative and Technical design features to enhance consumer appeal. Every product needs the packaging for protection from the external environment and needs the labelling to communicate with the external world. It also acilitates brand positioning and brand promotion.
Packaging and Labelling are complements to each other. Labelling has 3 basic elements – Text, Graphics & Substrate.
Packaging (primary, secondary and tertiary) decides the substrate and its specifications. Substrates provide the base for the texts and graphics. While the text is for the brand / product identity and statutory compliance, graphics is for the mproduct positioning or brand identity. Fonts, colors, embossing, hot stamping, different types of surface finish are common. Packaging and Labelling also contains many value added features to facilitate specific functions like ease in display, dispensing and disposal. Such features combinedly promote branding.
Tamper evident features
Design or device to prevent pilferage or theft of the product from the original pack. Tamper evidence is already an existing system, primarily to prevent theft and pilferage. There are 3 common systems currently being used for tamper evidence:
1) Use of tamper evident stickers, 2) Use of hot or cold glue, and 3) Mechanical locking of the pack
which leaves sign / proof when opened. It also has challenges like use of an additional sticker or glue and related application units on the packaging lines. Industry is using it for years to prevent theft or copying of the product-packs.
Anti-counterfeit features
Design or device which prevents to replicate or cloning of the original product-pack. Some of the common anti-counterfeit features on product-packaging are:
1. Overt Technologies (visible features): These are visible but difficult to imitate – customized holography, micro text, color changing security inks / film, security graphics, random serialization, on-product imaging or numbering.
2. Covert Technologies (invisible features): These are not visible with naked eyes – invisible printing, light sensitive printing, embedded images, digital watermark, anti-copy or anti scan design, laser codes.
3. Other features: RFID, biological taggants, micro taggants, secured web link, special (difficult to copy) printing / embossing on products (solid doses).
Authentication features
A tool or system to verify genuineness of the product and its ownership. This is the last action of an end-to-end solution wherein the end user participates to close the loop. Some of the common authentication features are intricate design stickers / holograms, microprinting, use of different electronic tags / chips, SMS based communication link, web link through Wi-Fi connectivity, biometric codes, speciöc light sensitive prints / codes, mobile application-based codes, scratch and scan codes, and so on. Product level authentication features are also available, especially on solid dose formulations.
These features are often used as a single technology or as a blend of
different technologies, involving other value-added features like, anti-counterfeiting, tamper
evidence or even serialization. However, in all these cases

customer education and awareness building is must through physical demonstrations or printed instructions.
Track and Trace features
A tool or system to control and ensure supply chain security and assure consumer safety. The Track and Trace feature was primarily introduced to monitor and control the movement of the goods across the entire supply chain. This GTIN based technology uses unique serial number on each trade unit / saleable pack. The GTIN and batch specific data (packaging level wise aggregated) is pushed to the network for verification and authentication by the regulators and agencies involved in the supply chain of the products. Despite initial challenges, it has been broadly accepted and adopted by pharma industry not only to ensure supply chain security, but also to control counterfeit, pilferage and diversion. Pharma industry across the globe is now aligned to make it a success with various cost effective, compact and compliant solutions.
Automation-Digitalization-Communication has influenced and improved every stage of operations in the industry and it has been adapted to our changing lifestyle as well.
- Evolution to Revolution: Local to Global – Large wholesale pack to Small consumer pack.
- Scaling Up: Quantity with Quality – Consistency and Reproducibility.
- Manufacturing and Packaging: Testing and Releasing – Data management and Documentation.
- Promotion and Protection: Cloning and Counterfeiting –Theft and Diversion.
- Storage, Handling and Transport: Logistics and Distribution – Safety, Security and Traceability.
- Digitalization is a great support to Automation and Communication systems through Data and Information Capture, Data Transform, Storage, Processing, Transfer, Exchange, Retrieval, Integrity, Transparency and Compliance.
- These help to optimize cycle time, space utilization and convenience. It enhances speed to all the business processes. Quality and Compliance are well ensured with Transparency and Integrity. It also strengthens the Communication system.
There are many basic tools to support this Automation-Digitalization-Communication relationship. Over the period these tools have been fine-tuned for their practical applicability. Such tools help to build different custom-made programs, which directly or indirectly facilitate the operations, conversions, controls and communications
This is how Product-Packaging-Labelling relationship is strengthened with the help of advanced tools and technologies. Often multiple tools and technologies are blended to get a customized hybrid model, which are more powerful than a single technology
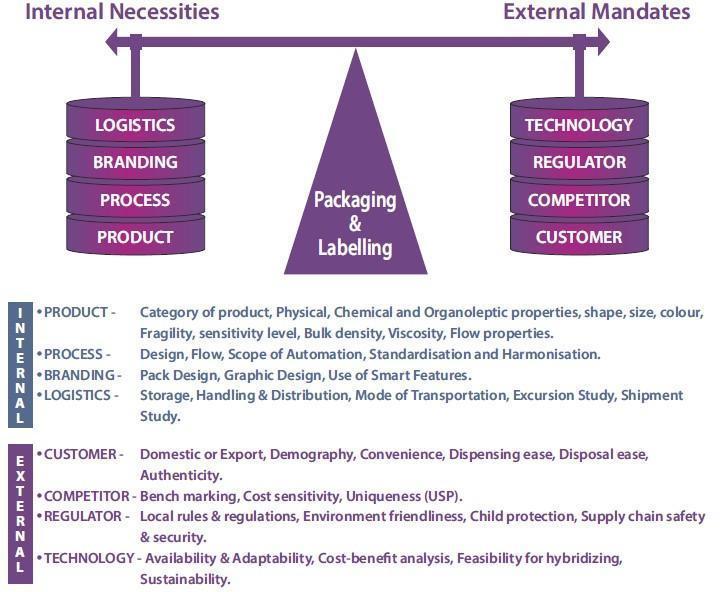
 Across the world packaging professionals always try to and customized solution to support brand protection and brand promotion with their knowledge in material science, conversion technologies, application feasibility and flexibility. Basic packaging materials vary from simple paper to complex composites, inclusive of metals, glass, plastics, and many more options. Accordingly, their conversion technologies also vary to önalize the design and process options. While doing so it is always ensured basic safety and security of the product-pack is maintained.
Across the world packaging professionals always try to and customized solution to support brand protection and brand promotion with their knowledge in material science, conversion technologies, application feasibility and flexibility. Basic packaging materials vary from simple paper to complex composites, inclusive of metals, glass, plastics, and many more options. Accordingly, their conversion technologies also vary to önalize the design and process options. While doing so it is always ensured basic safety and security of the product-pack is maintained.
There are logistics challenges as well which covers multiple options like roadways, railways, airways, ropeways, pipelines and water routes. Each of these has their own hazards and challenges. Similarly, there are many external mandates like compliance to regulatory guidelines, benchmarking with competitors, obsoletion of technology, change in consumer behaviors, etc. Hence a good balance is required while designing and developing a product-pack which can sustain and survive longer in competition.
 Prabir K Das, is one of the first batch alumni (1985 – 87) from Indian Institute of Packaging, Mumbai (India), and a 30+ years-experienced pharmaceutical packaging professional, currently taking care of the Indian operations for one of the large and renowned global pharmaceutical service providers based in US. He is actively involved in development, execution and post commercialisation life cycle management which involves technology transfer, new product-pack launch, packaging validation, change management, quality and COGS improvement projects, automation, standardization and harmonization projects, track and trace project, and any kind of innovative packaging development project considering patient safety and compliance.
Prabir K Das, is one of the first batch alumni (1985 – 87) from Indian Institute of Packaging, Mumbai (India), and a 30+ years-experienced pharmaceutical packaging professional, currently taking care of the Indian operations for one of the large and renowned global pharmaceutical service providers based in US. He is actively involved in development, execution and post commercialisation life cycle management which involves technology transfer, new product-pack launch, packaging validation, change management, quality and COGS improvement projects, automation, standardization and harmonization projects, track and trace project, and any kind of innovative packaging development project considering patient safety and compliance.













